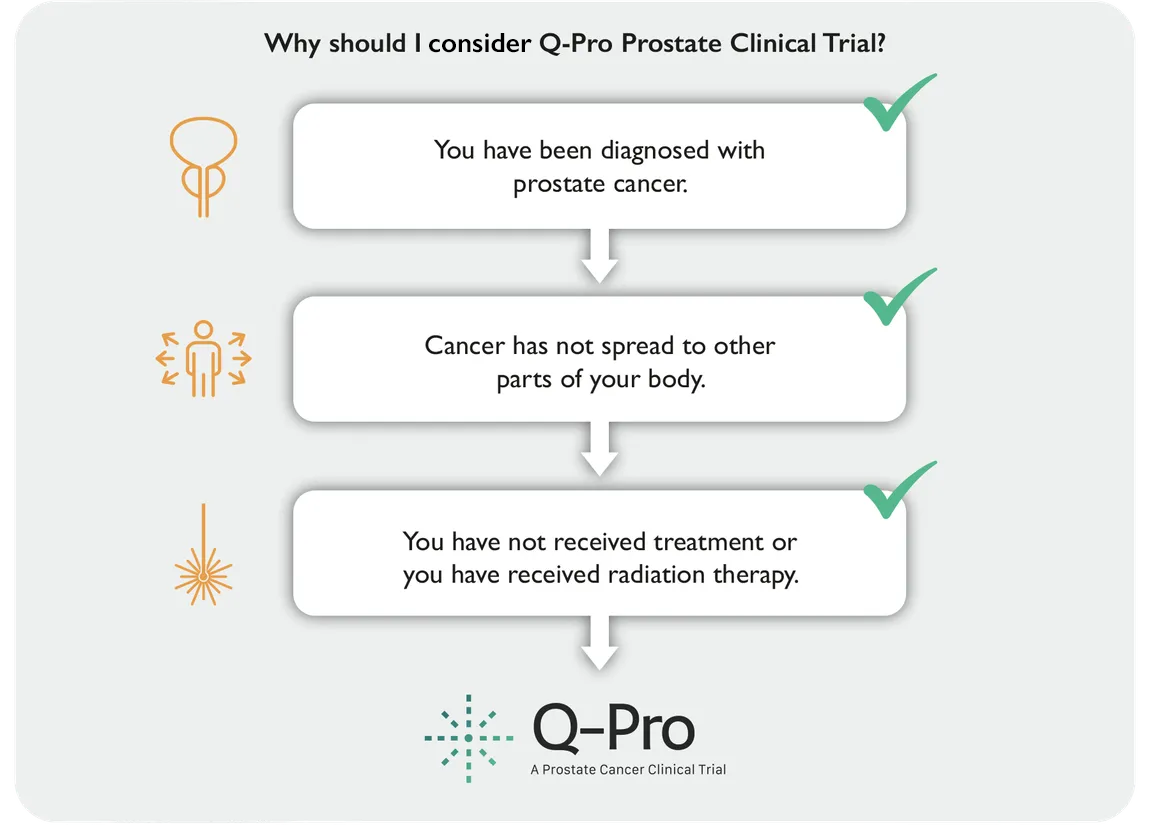
A new investigational method for the treatment of localized prostate cancer is currently available in two clinical trials. If you suffer from localized prostate cancer, there is a possibility to take part of the Q-Pro clinical trials and contribute to developing future cancer treatments.
A New Focal Approach to
Prostate Cancer Treatment
Prostate Cancer Treatment
The Q-Pro clinical trials evaluate a new focal treatment option for localised prostate cancer. The intervention is a focal and minimally invasive procedure. This means that the treatment targets only the tumour. The treatment is based on photodynamic therapy (PDT). The goal is to destroy the cancer cells while sparing surrounding healthy tissue. PDT is an approved treatment for some cancers that develop on the surface of the body, like skin cancer. The ambition is to provide an individualised treatment option that eliminates cancer from the prostate and minimises the side effects.
What you need to know
about the Q-Pro Clinical Trials
The Q-Pro clinical trials include one clinic visit for the study procedure. The procedure is performed during general anesthesia, and it begins with an injection of a drug that makes the tissue sensitive to light. Once a sufficient dose has reached the tumour, laser light is applied inside the prostate to activate the drug. Study participants may be going home the same day or stay in the hospital overnight. After the study procedure, participants will return to the clinic during the following year for study visits to check on their condition and monitor any side effects.
The drug used in the clinical trials is approved for treating eye diseases. The medical device used to deliver the treatment light individualises the therapy for each patient to optimise the destruction of cancer cells and reduce side effects.
See If You Are Eligible
If you're eligible, contact the clinic running the study. See contact details below.
Where is the Q-Pro Clinical Trial Taking Place?
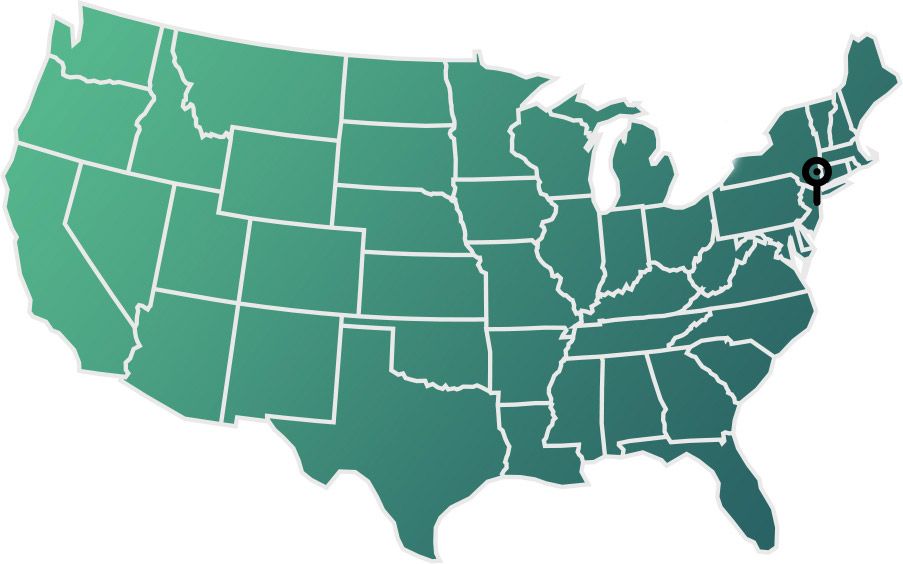
The study on recurrent prostate cancer is taking place at:
- Princess Margaret Cancer Centre in Toronto in Canada
- University College London Hospital in United Kingdom
- Memorial Sloan Kettering Cancer Center in New York in US
- Skåne University Hospital in Malmö in Sweden
Watch our videos to learn more about the two trials.
Frequently Asked Questions